
|
 |

Fully endoscopic vascular decompression of the trigeminal nerve
By Reza Jarrahy, M.D., Joseph Eby, M.D, Sung Tae Cha, M.D., Hrayr Shahinian MD, FACS
Summary
Microvascular decompression of the trigeminal nerve is an accepted and effective means of treating patients with trigeminal neuralgia in whom compression of the nerve by a vascular structure is implicated in the pathogenesis of the disease. The current standard technique uses the binocular operating microscope for all intra-operative visualization.
Posterior fossa endoscopy has demonstrated that the endoscope provides more comprehensive views of the anatomy of the cerebellopontine angle than does the operating microscope. To date, endoscopy has only been used to supplement microscopy in cranial nerve decompression surgery.
In this report, we describe our completely endoscopic surgical technique as we present the case of a patient with trigeminal neuralgia who underwent successful vascular decompression by this approach. Using this technique the offending vessel was separated from the nerve with minimal brain retraction or dissection of surrounding structures.
This report represents the first documented case where the endoscope was used as the exclusive imaging modality for decompression of the trigeminal nerve. From our experience we conclude that the endoscope's superior visualization more accurately identifies neurovascular conflicts, and provides a comprehensive evaluation of the completeness of the decompression. Additionally, this new method minimizes the risks of brain retraction and extensive dissection often required for microscopic exposure. From this study we conclude that completely endoscopic vascular decompression represents the next step forward in the safe and effective surgical treatment of trigeminal neuralgia.
Keywords
Endoscopy, Microvascular decompression, Trigeminal neuralgia
Introduction
Trigeminal neuralgia is a disease characterized by severe and often debilitating facial pain that occurs along the distribution of any of the three branches of the trigeminal nerve. As classically defined, attacks are intermittent and the quality of the pain is sharp, stabbing, or mimicking an electric shock. The mainstay of treatment for patients with trigeminal neuralgia prior to the modern surgical era was confined to either medical management of episodic symptoms, or sectioning of the trigeminal nerve.[1-4]
In 1934 Dandy first hypothesized that vascular compression of the trigeminal nerve by aberrant vessels of the posterior circulation may be responsible for this disorder.[4] Building upon Dandy's observations, Jannetta proposed what is our current understanding of the pathogenesis of trigeminal neuralgia when in 1967 he stated that compression and distortion of the root entry zone of the trigeminal nerve by aberrant venous and arterial structures was a causative factor in the etiology of trigeminal neuralgia.[5, 6] Jannetta also proposed that removal of these traumatic insults would result in alleviation of the neuropathic pain experienced by patients.[5, 6] These theories have been largely accepted, and have provided the basis for the current surgical management of trigeminal neuralgia. It was at this time Jannetta also popularized the use of the operating microscope for vascular decompression.[7] Over the past thirty years several large series reports have documented very good results for the microscopic decompression operation, with minimal morbidity and almost no mortality.[9-13] For this reason the microscopic decompression operation has become the gold standard surgical treatment for trigeminal neuralgia.
Rigid endoscopy, however, has emerged as an imaging tool that rivals the quality of visual data provided by the microscope in many neurosurgical applications.[14-17] Specific evidence that the combination of microscopy and endoscopy, relative to microscopy alone, provides superior visual information in cranial nerve decompression surgery has been offered.[18] In this report we describe the case of a patient with trigeminal neuralgia in whom rigid endoscopes were used as the sole means of intra-operative imaging during vascular decompression of the trigeminal nerve. To our knowledge, this case represents the first reported instance of an entirely endoscopic approach to this procedure.
Patients/Methods
History. This 66-year-old woman presented to our service upon the recommendation of her neurologist, reporting a 20-year history of right-sided tic douleureux. Her neuralgia was characterized by intermittent attacks of sharp pain along the distribution of all three branches of the trigeminal nerve. These episodes were often triggered by mastication, thereby restricting her diet to foods that caused the least amount of intraoral trauma during ingestion (e.g., liquids, puddings, purees). Although the patient stated that she did achieve satisfactory pain relief with her medical regimen, which included elevated doses of Carbamazepine and Baclofen, she complained that the side effects of the medication (most notably the resultant somnolence and dysarthria) significantly diminished the quality of her daily life.
Thin section T1- and T2-weighted magnetic resonance (MR) images of the base of the skull and CPA were obtained, in addition to magnetic resonance angiography (MRA) of the regional vascular anatomy. While no discreet vessel was noted to be compressing the right trigeminal nerve, there was a suggestion of vessel aberrance in the prepontine cistern. After being fully informed regarding the overall risks and benefits of trigeminal nerve decompression, as well as the particular benefits associated with the use of endoscopy in lieu of the microscope, the patient consented to surgery.
Operation. Following the induction of general anesthesia, the patient was placed in the park bench position. The neck was flexed and the head was slightly rotated to the right before being fixed in a Mayfield head clamp. A 3 cm retroauricular curvilinear incision was made and carried down to bone. Subperiosteal musculocutaneous flaps were elevated anteriorly and posteriorly and then retracted, allowing for a 2.0 cm diameter craniotomy to be made at the confluence of the transverse and sigmoid sinuses. Dura was exposed and incised (Figure 1), and cerebrospinal fluid (CSF) was drained. A 4.0 mm 0° rigid endoscope (Karl Storz of America, Culver City, CA) was introduced into the posterior fossa and was stabilized in place with the use of a pneumatically powered holding arm (Mitaka Kohki Co., Tokyo) (Figure 2). It was advanced to the level of the trigeminal nerve, and survey of the regional neurovascular anatomy was performed, revealing a looping branch of the superior cerebellar artery (SCA) compressing the inferior surface of the nerve's root entry zone. Dissecting instruments were used to liberate the nerve from the vessel (Figure 3). Once this was achieved, two pads of Teflon were interposed between the nerve and vessel to maintain the separation (Figure 4). A final survey utilizing the 300endoscope was conducted to evaluate the adequacy of the decompression. As no further intervention was required, a fibrin-based sealant (Hemaseel APR™, Haemacure Co., Sarasota, FL) was applied over the Teflon pledgets, the dura was closed, the bone flap repositioned, and the skin flaps re-approximated.
Results
The patient awoke from the surgery free of pain, and was monitored in the intensive care unit overnight without any demonstration of neurological deficit. Her postoperative course was unremarkable except for insomnia and irritability consistent with acute anticonvulsant withdrawal. She was discharged from the hospital on the fourth postoperative day and was tapered off of Carbamazepine over the subsequent week. She remains pain free without the use of any medication six months following the procedure.
Discussion:
The concept of using endoscopes to visualize the anatomy of the posterior fossa is not new. In 1917, Doyen described a technique for trigeminal root neurectomy under endoscopic guidance via an occipital craniotomy.[3] Since then, published experience with posterior fossa endoscopy has been scarce. Isolated reports detailing endoscopic exploration of the posterior fossa for diagnostic purposes and for selective trigeminal nerve root sectioning were published in the 1970s, but only during the past decade has interest in the application of endoscopic imaging to this region been renewed.[19, 20] In 1992 O'Donoghue and O'Flynn described and classified the endoscopic anatomy of the CPA.[21] Since then, a number of neurotologists have presented their experiences with CPA endoscopy, particularly as an imaging modality in acoustic neuroma surgery.[22-28] A recently published atlas specifically details the endoscopic surgical anatomy of the cranial nerves at the posterior skull base. [29]
Much of the impetus driving the "renaissance" in posterior fossa endoscopy as well as minimally invasive surgical practice in general lies in the recent development of appropriate endoscopic instrumentation.[30] The latest generation of endoscopic instrumentation offers devices that are specifically tailored to intracranial application. These include Hopkins rod telescopes of varying lengths and diameters, lens systems that allow viewing over a wide range of angles, various illumination devices, digital video recorders and processors, sturdy endoscope holding arms, and endoscope irrigation sheaths.[18, 29-32]
The key to successful vascular decompression surgery is accurate intra-operative visualization of all nerve-vessel conflicts and verification of complete nerve decompression.[8-10] A recent review article of over 2700 patients undergoing microvascular decompression for trigeminal neuralgia documented an overall 6-38% failure rate and a 20-25% recurrence rate.[13] Studies of patients re-explored for persistent or recurrent symptoms demonstrate that between 10-86%are found to have a new area of vascular compression.[8, 33-37] Some authors have attributed these results to incomplete identification of all nerve-vessel conflicts or to incomplete decompression of the nerve at the time of initial surgery.[8, 33-38]
The strength of the endoscope in this setting is its ability to impart to the surgeon more comprehensive anatomical images of the trigeminal nerve and surrounding structures than those obtainable under the static anteroposterior viewing capabilities of the microscope. Spencer recently documented the superior visualization provided by even the 00 endoscope, wherein he found a 1.5-2.5 times greater volume of view compared to the operating microscope.[39] Magnan in a recent series of 60 patients with hemifacial spasm used the endoscope to supplement microscopic decompression and noted and additional 72% accuracy in identifying nerve-vessel conflicts.[18] A similar study performed by our group on 21 patients undergoing endoscope-assisted microvascular decompression of the trigeminal nerve demonstrated that 27% of conflicts were missed by microscopic exploration alone, and 24% of microscopic decompressions were found to be incomplete.[40]
These early results suggest that the use of endoscopy for vascular decompression surgery may reduce the number of initial failures and recurrences when compared to the microscopic approach. In the case presented above, the exclusive use of the endoscope provided a more accurate means of identifying all implicated nerve-vessel conflicts, while the addition of the 300angled endoscope was able to confirm complete decompression.
During our endoscopic procedure we also noted a marked advantage of the endoscope in gaining access to the cerebellopontine angle, as we were able to enter this region with minimal dissection or retraction. This is in contrast to the microscope's straight line of sight view, which is frequently obscured by the narrow tortuous confines of this region, often requiring significant dissection and retraction for adequate exposure. Serious complications as a result of vascular decompression surgery (other than cranial nerve deficits) are most frequently related to brain retraction injury, and are estimated to occur in approximately 10% of skull base surgical procedures.[41] We believe that less invasive exposure of this region using the endoscopic technique will result in future reports documenting a lower incidence of complications.
The endoscopic method is not without some difficulties, many of which have been overcome by the advances in technology such as the rigid endoscope holding arm and irrigation sheath mentioned previously. Other aspects of this approach also require some technical adjustment for the surgeon, primarily in the manipulation of surgical instrumentation in a three-dimensional field, while monitoring the procedure on a two dimensional screen.
This report documents the first case of vascular decompression of the trigeminal nerve performed entirely endoscopically. We found the endoscope offers several advantages over the operating microscope, including atraumatic access to the cerebellopontine angle, improved visualization of all neurovascular conflicts, as well as a more comprehensive evaluation of the adequacy of decompression. All of these beneficial aspects of endoscopy should have direct implications for improved surgical outcome. Leading us to conclude that fully endoscopic vascular decompression represents the next step forward in the safe and effective surgical treatment of trigeminal neuralgia.
References
- Blitzer, A., Pharmacotherapy and trigeminal neuralgia. Anesth Prog, 1969. 16(1): p. 15-7 passim.
- Tovi, D., D.M. Leivy, and E. Krueger, Trigeminal neuralgia. Current trends in treatment. N Y State J Med, 1969. 69(4): p. 564-7.
- Doyen, E., Surgical therapeutics and operative techniques,, Balliere, Tindall, and Cox, Editors. 1917: London. p. 599-602.
- Dandy, W., Concerning the cause of trigeminal neuralgia. American Journal of Surgery, 1934. 24: p. 447-55.
- Jannetta, P.J., Arterial compression of the trigeminal nerve at the pons in patients with trigeminal neuralgia. J Neurosurg, 1967. 26(1): p. Suppl:159-62.
- Jannetta, P.J., Gross (mesoscopic) description of the human trigeminal nerve and ganglion. J Neurosurg, 1967. 26(1): p. Suppl:109-11.
- Jannetta, P.J., The surgical binocular microscope in neurological surgery. Am Surg, 1968. 34(1): p. 31-4.
- Jannetta, P.J. and D.J. Bissonette, Management of the failed patient with trigeminal neuralgia. Clin Neurosurg, 1985. 32: p. 334-47.
- Barker, F.G., 2nd, et al., The long-term outcome of microvascular decompression for trigeminal neuralgia [see comments]. N Engl J Med, 1996. 334(17): p. 1077-83.
- McLaughlin, M.R., et al., Microvascular decompression of cranial nerves: lessons learned after 4400 operations [see comments]. J Neurosurg, 1999. 90(1): p. 1-8.
- Bederson, J.B. and C.B. Wilson, Evaluation of microvascular decompression and partial sensory rhizotomy in 252 cases of trigeminal neuralgia. J Neurosurg, 1989. 71(3): p. 359-67.
- Cutbush, K. and R.L. Atkinson, Treatment of trigeminal neuralgia by posterior fossa microvascular decompression. Aust N Z J Surg, 1994. 64(3): p. 173-6.
- Lovely, T.J. and P.J. Jannetta, Microvascular decompression for trigeminal neuralgia. Surgical technique and long-term results. Neurosurg Clin N Am, 1997. 8(1): p. 11-29.
- Fries, G. and A. Perneczky, Endoscope-assisted brain surgery: part 2--analysis of 380 procedures. Neurosurgery, 1998. 42(2): p. 226-31; discussion 31-2.
- Hopf, N.J. and A. Perneczky, Endoscopic neurosurgery and endoscope-assisted microneurosurgery for the treatment of intracranial cysts. Neurosurgery, 1998. 43(6): p. 1330-6; discussion 6-7.
- Wackym, P.A., et al., Endoscope-assisted vestibular neurectomy [see comments]. Laryngoscope, 1998. 108(12): p. 1787-93.
- Wackym, P.A., W.A. King, and D.J. Weisz, Vascular compression of the cochlear nerve identified by endoscopy during acoustic neuroma surgery. Otolaryngol Head Neck Surg, 1999. 120(4): p. 535.
- Magnan, J., et al., Hemifacial spasm: endoscopic vascular decompression. Otolaryngol Head Neck Surg, 1997. 117(4): p. 308-14.
- Oppel, F. and G. Mulch, Selective trigeminal root section via an endoscopic transpyramidal retrolabyrinthine approach. Acta Neurochir Suppl, 1979. 28(2): p. 565-71.
- Prott, W., [Cisternoscopy of the cerebellopontine angle (author's transl)]. Hno, 1974. 22(11): p. 337-41.
- O'Donoghue, G.M. and P. O'Flynn, Endoscopic anatomy of the cerebellopontine angle. Am J Otol, 1993. 14(2): p. 122-5.
- Bottrill, I.D. and D.S. Poe, Endoscope-assisted ear surgery. Am J Otol, 1995. 16(2): p. 158-63.
- Jennings, C.R. and G.M. O'Donoghue, Posterior fossa endoscopy [editorial]. J Laryngol Otol, 1998. 112(3): p. 227-9.
- King, W.A. and P.A. Wackym, Endoscope-assisted surgery for acoustic neuromas (vestibular schwannomas): early experience using the rigid Hopkins telescope. Neurosurgery, 1999. 44(5): p. 1095-100; discussion 100-2.
- Rosenberg, S.I., Endoscopic otologic surgery. Otolaryngol Clin North Am, 1996. 29(2): p. 291-300.
- Tatagiba, M., C. Matthies, and M. Samii, Microendoscopy of the internal auditory canal in vestibular schwannoma surgery. Neurosurgery, 1996. 38(4): p. 737-40.
- Magnan, J., et al., Surgical perspectives of endoscopy of the cerebellopontine angle. Am J Otol, 1994. 15(3): p. 366-70.
- Magnan, J., et al., Endoscopy of the cerebellopontine angle. Rev Laryngol Otol Rhinol, 1995. 116(2): p. 115-8.
- Magnan, J. and M. Sanna, Endoscopy in neuro-otology. Vol. 1. 1999, New York: Thieme. 7-23.
- Linder, T.E., D. Simmen, and S.E. Stool, Revolutionary inventions in the 20th century. The history of endoscopy [see comments]. Arch Otolaryngol Head Neck Surg, 1997. 123(11): p. 1161-3.
- Eaves, F.F., 3rd, J. Bostwick, 3rd, and F. Nahai, Instrumentation and setup for endoscopic plastic surgery. Clin Plast Surg, 1995. 22(4): p. 591-603.
- Nishioka, N.S. and M.A. Mycek, Initial experience with a real-time video processor for enhancing endoscopic image contrast. Gastrointest Endosc, 1998. 48(1): p. 62-6.
- Liao, J.J., et al., Reoperation for recurrent trigeminal neuralgia after microvascular decompression. Surg Neurol, 1997. 47(6): p. 562-8; discussion 8-70.
- Lee, S.H., et al., Recurrent trigeminal neuralgia attributable to veins after microvascular decompression. Neurosurgery, 2000. 46(2): p. 356-61; discussion 61-2.
- Kureshi, S.A. and R.H. Wilkins, Posterior fossa reexploration for persistent or recurrent trigeminal neuralgia or hemifacial spasm: surgical findings and therapeutic implications. Neurosurgery, 1998. 43(5): p. 1111-7.
- Yamaki, T., et al., Results of reoperation for failed microvascular decompression. Acta Neurochir, 1992. 115(1-2): p. 1-7.
- Rath, S.A., H.J. Klein, and H.P. Richter, Findings and long-term results of subsequent operations after failed microvascular decompression for trigeminal neuralgia. Neurosurgery, 1996. 39(5): p. 933-8; discussion 8-40.
- Klun, B., Microvascular decompression and partial sensory rhizotomy in the treatment of trigeminal neuralgia: personal experience with 220 patients. Neurosurgery, 1992. 30(1): p. 49-52.
- Spencer, W.R., et al., Approaches to the sellar and parasellar region: anatomic comparison of the microscope versus endoscope. Laryngoscope, 1999. 109(5): p. 791-4.
- Jarrahy, R., G. Berci, and S. HK, Endoscope-assisted microvascular decompression of the trigeminal nerve. Otolaryngol Head Neck Surg, 2000: p. Accepted for publication.
- Andrews, R.J. and J.R. Bringas, A review of brain retraction and recommendations for minimizing intraoperative brain injury [see comments]. Neurosurgery, 1993. 33(6): p. 1052-63; discussion 63-4.
Legend
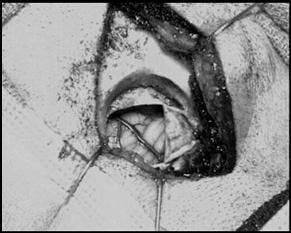
FIGURE 1. A 2.0-cm diameter posterior fossa craniotomy is made at the confluence of the sigmoid and transverse sinuses, and the dura is incised and reflected revealing cerebellum.
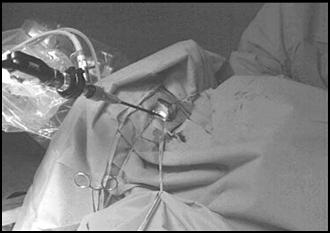
FIGURE 2. The 4.0 mm 0o rigid endoscope is introduced into the posterior fossa craniotomy and stabilized in place with the use of a pneumatically powered holding arm.
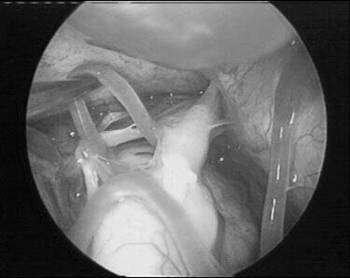
FIGURE 3. An intraoperative photograph taken from the endoscope reveals a looping branch of the superior cerebellar artery in close proximity to the trigeminal nerve. The trigeminal nerve is seen as it enters Meckel's cave.
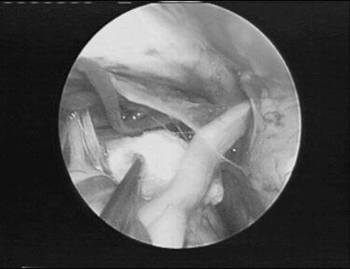
FIGURE 4. Pads of Teflon are interposed between the superior cerebellar artery and the trigeminal nerve.

|
 |
|
 |
|



